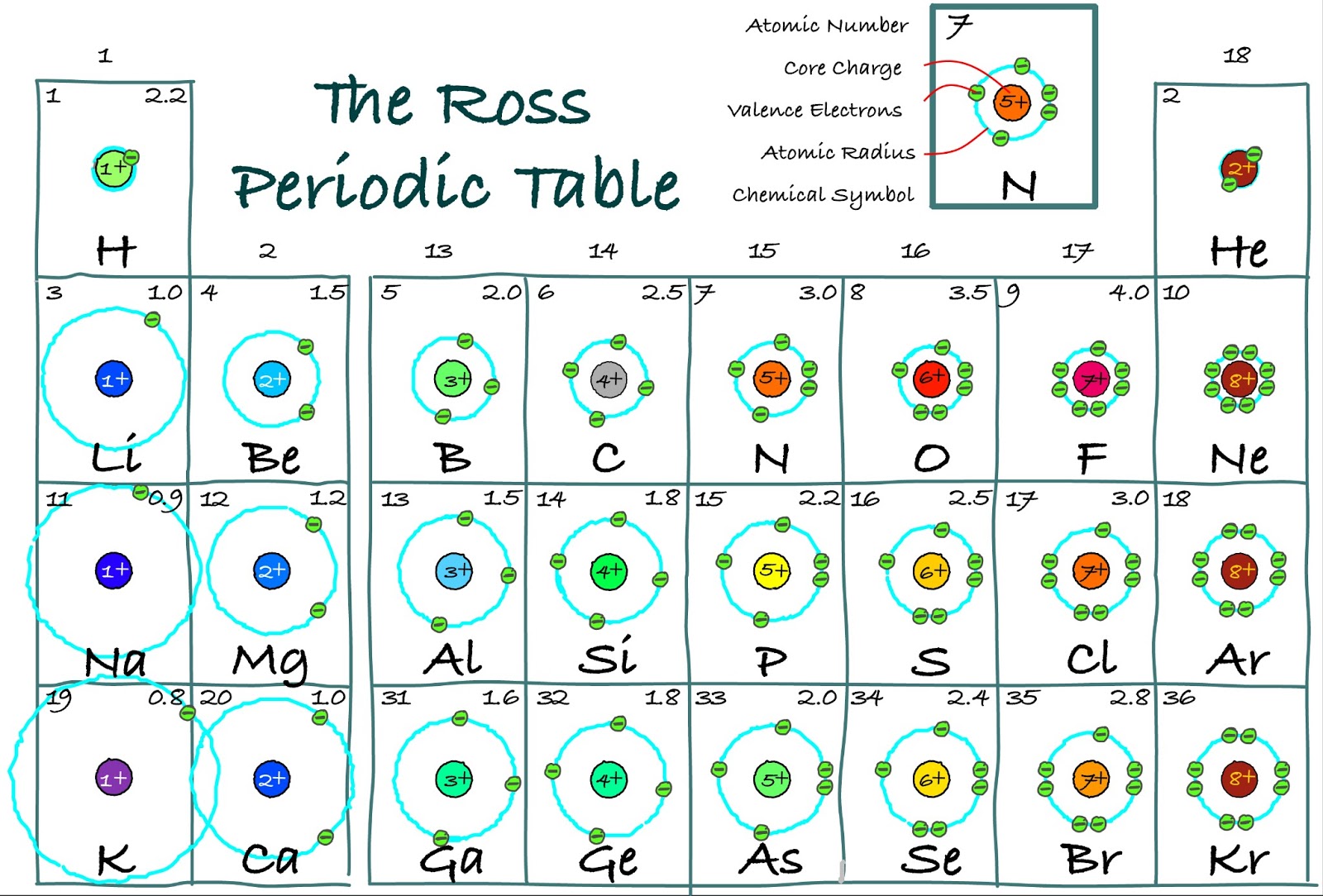
Based upon distributions across the periodic table of valence numbers, electron numbers, coordination numbers, and attached atom data suggestions are made regarding the placement of hydrogen in the periodic table, membership of group 3 in the periodic table, locations of the early actinoids in the periodic table, and assignments of certain elements as metalloids. These properties often display clear periodicity. Users may explore distributions of these classifications by percentages and heat map displays for individual elements or sets of elements, or in several cases for one classification plotted against a second. Its software calculates for an atom of interest within each compound classifications including valence number, oxidation number, coordination number, electron number, several covalent bond classifications, and the attached atom set. Because it is a noble gas, radon is not chemically reactive.The paper introduces Chemdex, a freely accessible web-based database of over 70 000 compounds characterised by crystallography from across the periodic table. Radon is an invisible, odorless noble gas that is slowly released from the ground, particularly from rocks and soils whose uranium content is high. For example Let’s take an example of a magnesium element. Atoms share electrons and form covalent bonds to satisfy the octet rule. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. In the second period elements, the two electrons in the 1s 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. The atoms in group 6A make two covalent bonds. Valence electrons are the electrons in the highest occupied principal energy level of an atom. For example, the elements of Group 1 are known as the alkali metals, Group 2 are the alkaline earth metals, Group 17 are the halogens, and Group 18 are the noble gases. Covalent bonds are formed by two atoms sharing electrons. In chemistry, valence electrons are the electrons in the outside or valence electron shell of an atom.They determine the valency of the atom which is important in how a chemical element reacts with other elements. Chemists often make general statements about the properties of the elements in a group using descriptive names with historical origins. However, even these small quantities are required for the body to function properly.Īs previously noted, the periodic table is arranged so that elements with similar chemical behaviors are in the same group. 
These last three metals are not listed explicitly in Table 2.1.2, so they are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. The elements in a group have similar physical or chemical characteristics of the outermost electron. There are 18 numbered groups in the periodic table the 14 f-block columns, between groups 2 and 3, are not numbered. Manganese is needed for the body to metabolize oxygen properly. In chemistry, a group (also known as a family) 1 is a column of elements in the periodic table of the chemical elements. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
